Zircon
Zircon
Zirconium is a chemical element (symbol Zr). Zirconium is a non-toxic heavy metal in the titanium group of the periodic table.
Zircon (also referred to as zirconium silicate), ZrSiO4, is a naturally occurring mineral found in many colours, which is used a gem; the colourless version is sometimes used as a diamond substitute. Chemically it is a mixture of varying ratios of silicon dioxide (SiO2) and zirconium dioxide (ZrO2), generally with hafnium. Zircon sand is used as the raw material for manufacturing zirconium dioxide.
Zirconia (ZrO2) is a transparent diamond imitation gem, manufactured synthetically from zirconium dioxide. More precisely it is CSZ, cubic stabilised zirconium dioxide at very high temperatures (above 2370°C) due to the addition of other metal oxides. When stabilisation involves using yttrium oxide the term is YSZ and with calcium oxide it is CaSZ.
Zirconium dioxide (ZrO2), also zirconium(IV) oxide, zircon oxide, (previously also referred to as zircon acid or zirkonerde [zirconia]) is a high performance oxide-ceramic. The naturally occurring mineral in monoclinic (at room temperature to 1173°C crystallising) form (modification) is called baddeleyite.
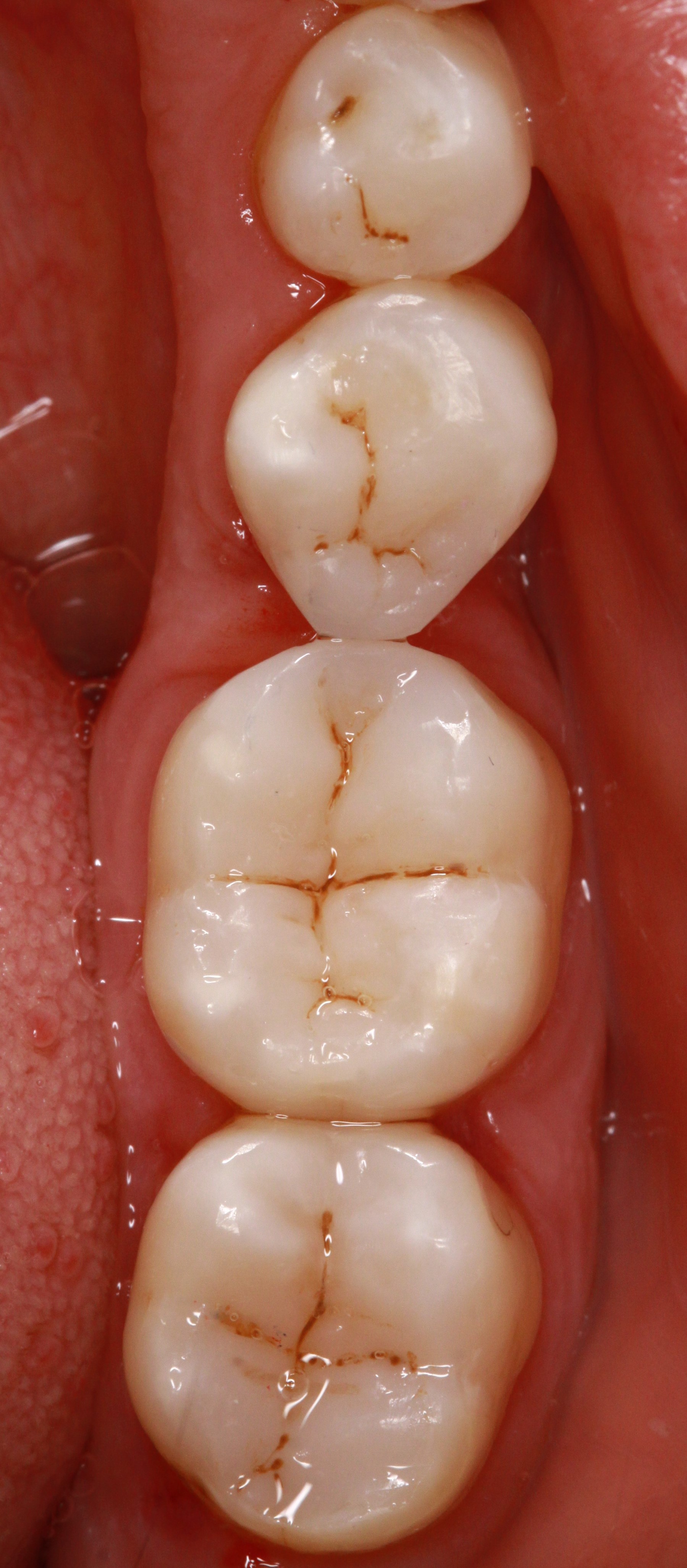
 All-zirconia bridgework and crowns
All-zirconia bridgework and crowns
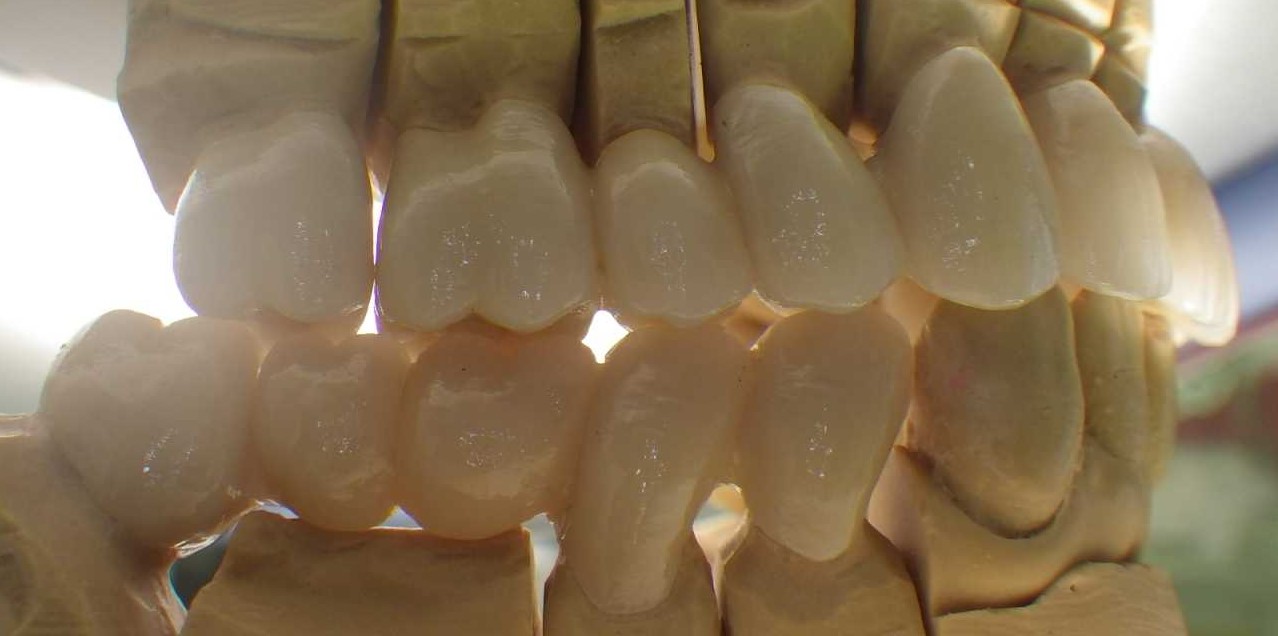
 Zirconium dioxide free-end bridge
Zirconium dioxide free-end bridge
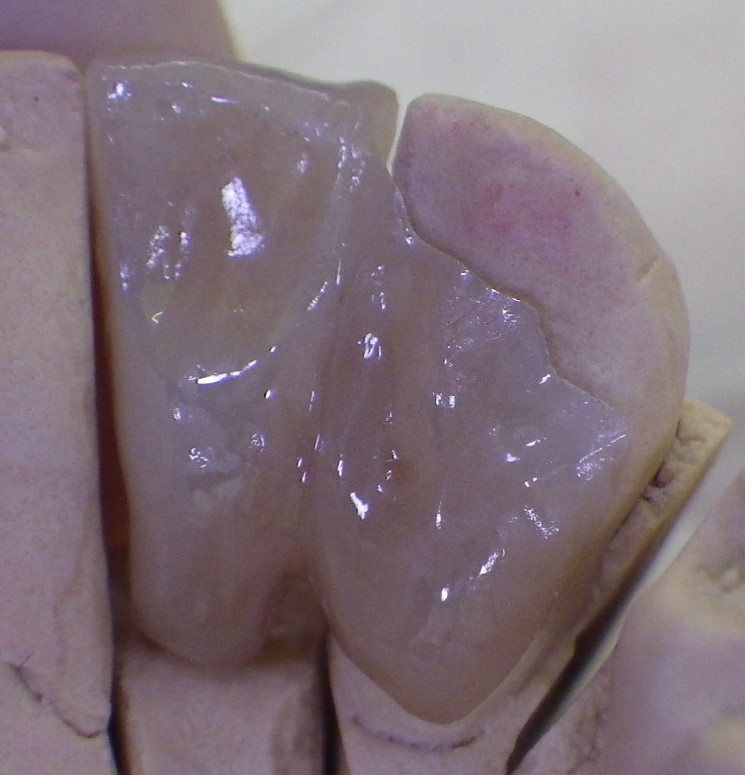 Lingual view of single-winged bridge
Lingual view of single-winged bridge
Compaction of highly pure zirconium dioxide powder and binding agents without heat treatment produces a press blank, known as a green compact or green body. Shrinkage of 25% at a later stage must be taken into account. A presintering process is then completed at approx. 1000°C, during which the binding agents are eliminated, shrinkage of approx. 5% occurs. This produces a partially sintered blank. In this partially sintered state the zirconium dioxide can be processed using many conventional dental CAD-CAM milling systems. Subsequent shrinkage of 20% at the (post) sinter firing stage must be factored into the calculation by respective enlarged manufacturing.
Alternatively, the zirconium dioxide can also first be densely sintered and then hot isostatically pressed (recompacted). The resultant material undergoes no further shrinkage and can therefore be processed dimensionally accurately (1:1), however, this material is extremely hard and the tool wear exceptionally high.
CeSZ zirconium dioxide partially or fully stabilised with cerium oxide.
CaSZ zirconium dioxide stabilised with calcium, form of zirconia.
CSZ cubic stabilised zirconium dioxide (above 2370°C) TSZ tetragonal stabilised zirconium dioxide (temperature range 1173°C to 2370°C)
YSZ zirconium dioxide partially or fully stabilised with yttrium oxide.
Want to give it a try ...
... or need professional advice?
Get in touch with us or click Contact.
Word of the day
| English | German |
|---|---|
| surrounding | umgebend, umliegend |
Focus text of the month
Composites also composite (from the Latin componere = to compose) are tooth-coloured filling materials with plastic properties used in dental treatment. In lay terms they are often referred to as plastic fillings, also erroneously sometimes confused with ceramic… Composites also composite (from the Latin componere = to compose) are tooth-coloured filling materials with plastic properties used in dental treatment. In lay terms they are often referred to as plastic fillings, also erroneously sometimes confused with ceramic fillings due to their tooth colour. After being placed in a cavity they cure chemically or by irradiating with light or a combination of the two (dual-curing). Nowadays, composites are also used as luting materials. The working time can be regulated with light-curing systems, which is a great advantage both when placing fillings and during adhesive luting of restorations. Dual-curing luting materials are paste/paste systems with chemical and photosensitive initiators, which enable adequate curing, even in areas in which light curing is not guaranteed or controllable. Composites were manufactured in 1962 by mixing dimethacrylate (epoxy resin and methacrylic acid) with silanized quartz powder (Bowen 1963). Due to their characteristics (aesthetics and advantages of the adhesive technique) composite restorations are now used instead of amalgam fillings.
The material consists of three constituents: the resin matrix (organic component), the fillers (inorganic component) and the composite phase. The resin matrix mainly consists of Bis-GMA (bisphenol-A-glycidyldimethacrylate). As Bis-GMA is highly viscous, it is mixed in a different composition with shorter-chain monomers such as, e.g. TEGDMA (triethylene glycol dimethacrylate). The lower the proportion of Bis-GMA and the higher the proportion of TEGDMA, the higher the polymerisation shrinkage (Gonçalves et al. 2008). The use of Bis-GMA with TEGDMA increases the tensile strength but reduces the flexural strength (Asmussen & Peutzfeldt 1998). Monomers can be released from the filling material. Longer light-curing results in a better conversion rate (linking of the individual monomers) and therefore to reduced monomer release (Sideriou & Achilias 2005) The fillers are made of quartz, ceramic and/ or silicon dioxide. An increase in the amount of filler materials results in decreases in polymerisation shrinkage, coefficient of linear expansion and water absorption. In contrast, with an increase in the filler proportion there is a general rise in the compressive and tensile strengths, modulus of elasticity and wear resistance (Kim et al. 2002). The filler content in a composite is also determined by the shape of the fillers.
Composite restorations Conclusion |

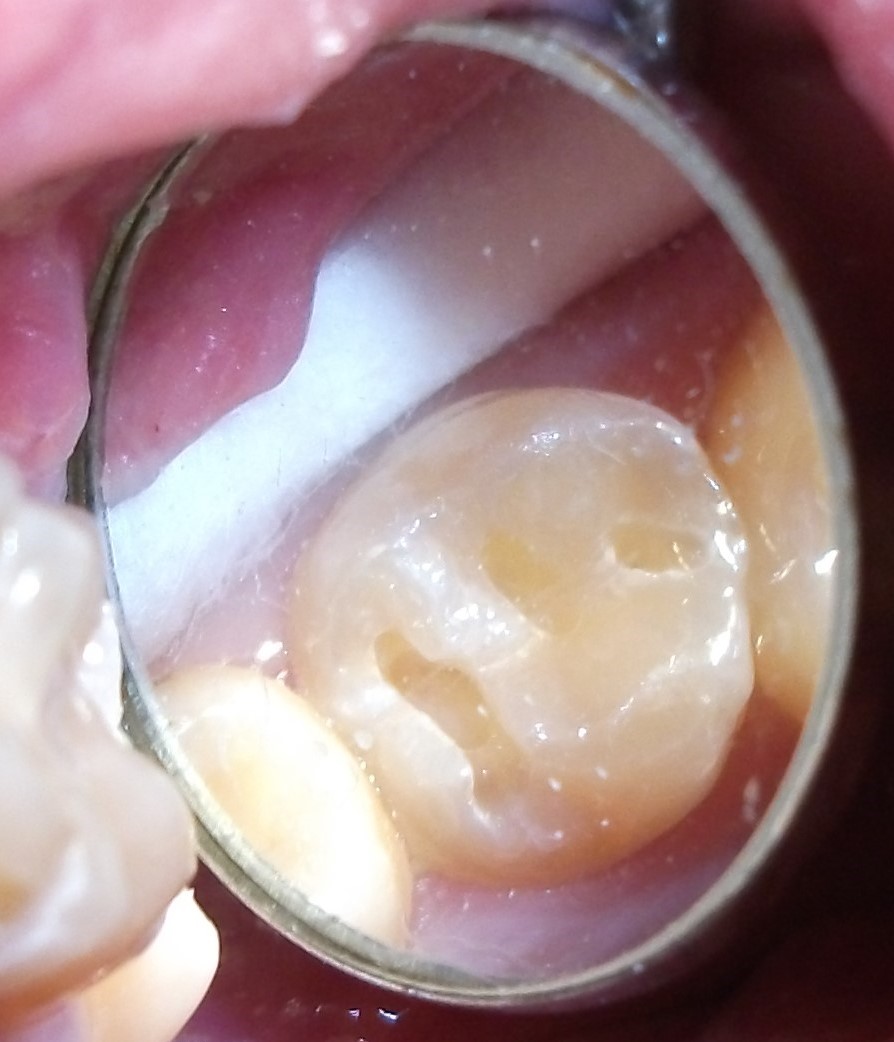 Minimally-invasive preparation and
Minimally-invasive preparation and 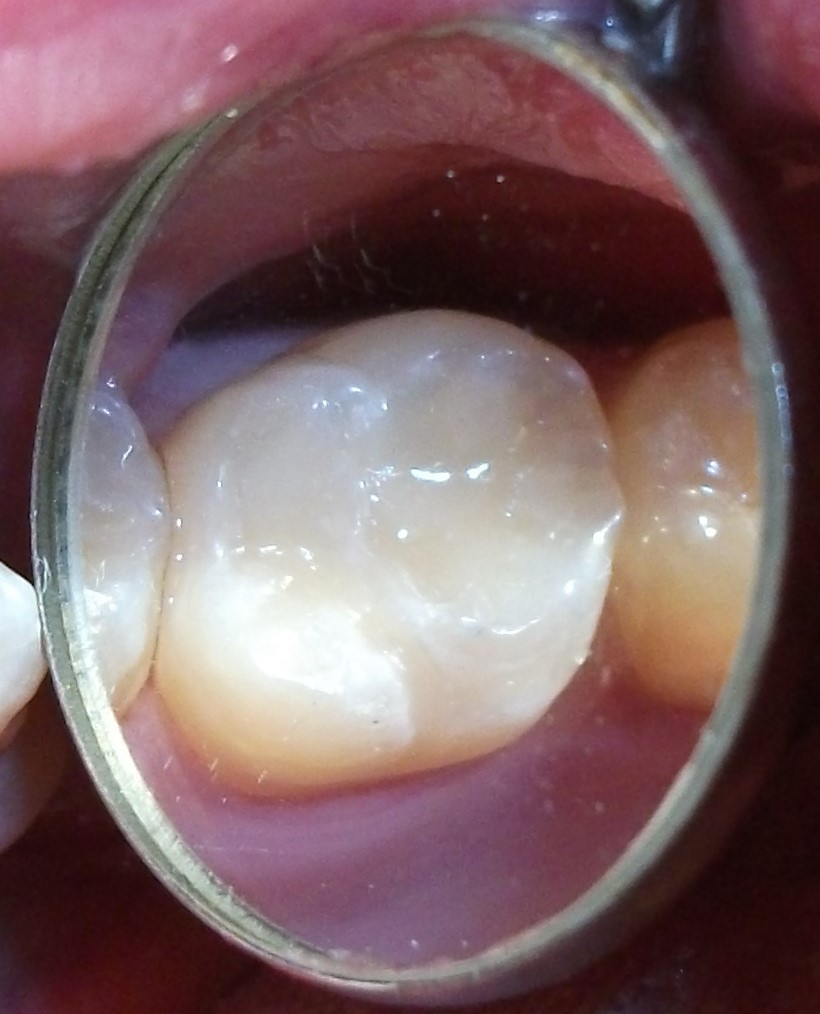 indiscernible composite restoration
indiscernible composite restoration