Air-abrasion units in dentistry
Air-abrasion units in dentistry
Air-abrasion units attached to standard handpiece couplings of dental units (or as stand-alone units) are always operated in dental medicine (unlike the sandblasters in dental technology) using a mixed jet of downstream separated (avoids clumping) water, compressed air and powder. The powder is stored in a refillable reservoir ("powder chamber") in the unit.
Introduced as far back as the mid-20th century, air-abrasion units have again been used since about the year 2000 ("cavity air polishers") operating at a pressure of between 4 and 10 bar for reducing hard tooth structure (air abrasion, "kinetic cavity preparation", abbreviated as "KCP"). The impact of water-insoluble aluminium oxide particles (equivalent to dental technological "abrasive grit") prepares non-contact (no tactile sensation), smaller, minimally invasive cavities quietly and with only low pain, without generating heat (unlike rotary instruments). The cavities, however, are not prepared with predictable geometries (undefined margins). Other areas of application are the intensive cleaning of fissures before sealing and conditioning (roughening, creation of micro-retention and/or chemical changes) of restorations or tooth surfaces for improving the bond strength, particularly of adhesive systems (also to veneering porcelains, e.g. for intraoral repairs).
The more widely used powder/water jet units (since approx. 1980) operate at a pressure of 2 to 3 bar with angular crystals made from water-soluble, very salty tasting, sodium hydrogen carbonate (NaHCO3, "natron"; obsolete also "sodium bicarbonate") powder (due to "refined" hydrophobic substances), later also reputedly less abrasive, water-insoluble "floury" calcium carbonate (CaCO3). They are used for targeted supragingival removal of discoloration from enamel (professional cosmetic teeth cleaning).
The classically used "salt jet" can cause damage to adjacent restoration surfaces, exposed dentine surfaces or injury to the gingiva. Applied at an unfavourable angle, the jet of air can lead to gingival emphysema, in the worst-case scenario later to air embolisms. When using air-abrasion units, special safety precautions should also be taken to avoid germ transfer, blocked tubing, damage to objects, inhalation or eye injuries.
In recent years, water-soluble amino acid glycine has been increasingly used for "perio-polishing", i.e. for subgingival removal of accretion and bacteria from the gingival pockets for prophylactic and periodontal therapy purposes. In the absence of calculus, this procedure is deemed superior to scaling with instruments.

Guided Biofilm Therapy (GBT) is a synonym for systematic prophylaxis and guarantees a uniformly high standard of quality in oral medical prevention, prophylaxis and therapy. GBT unites the most important scientific experience with technically state-of-the-art instruments – minimally invasive yet maximally preventive – all in a single holistic treatment session.
Want to give it a try ...
... or need professional advice?
Get in touch with us or click Contact.
Word of the day
| English | German |
|---|---|
| plaster solvent | Gipsauslöser, Gipsentferner, Gipslösebad, Gipslösemittel, Gipslöser |
Focus text of the month
Root canal posts are inserted centrally into adequately long, straight sections of devitalised, root-filled teeth which show no pathological findings either clinically or radiologically. Severe vertical and horizontal structure loss from the clinical crown is an indication… Root canal posts are inserted centrally into adequately long, straight sections of devitalised, root-filled teeth which show no pathological findings either clinically or radiologically. Severe vertical and horizontal structure loss from the clinical crown is an indication for root canal posts. The preferred method is generally to insert the root canal post, so that it (by predrilling with ascending diameters) virtually fills the prepared post site and fits close to the dentine wall. That section of the root canal post in the root should be at least as long as the crown restoration, leaving a few millimetres of the root canal filling to seal the apex. Isolated concepts have also been developed with smooth metal posts projecting apically beyond the tooth and intended for anchoring in the bone (transdental fixation).
Restoration with a temporary post is seldom practical due to the increased fracture and reinfection rate; a definitive root canal post should be inserted as early as possible.
Customised root canal posts (often made from precious metal alloys), cast after waxing up directly in the root canal or indirectly after taking an impression of the root canal lumen (using burnout posts), can also be used for filling lumina with oval or irregular cross sections, as well as two or three divergent canals each fitted with a post inserted, if necessary, through the opening of another canal. The core build-up(s), or even the entire crown (historically: post crown) can be integrated. |

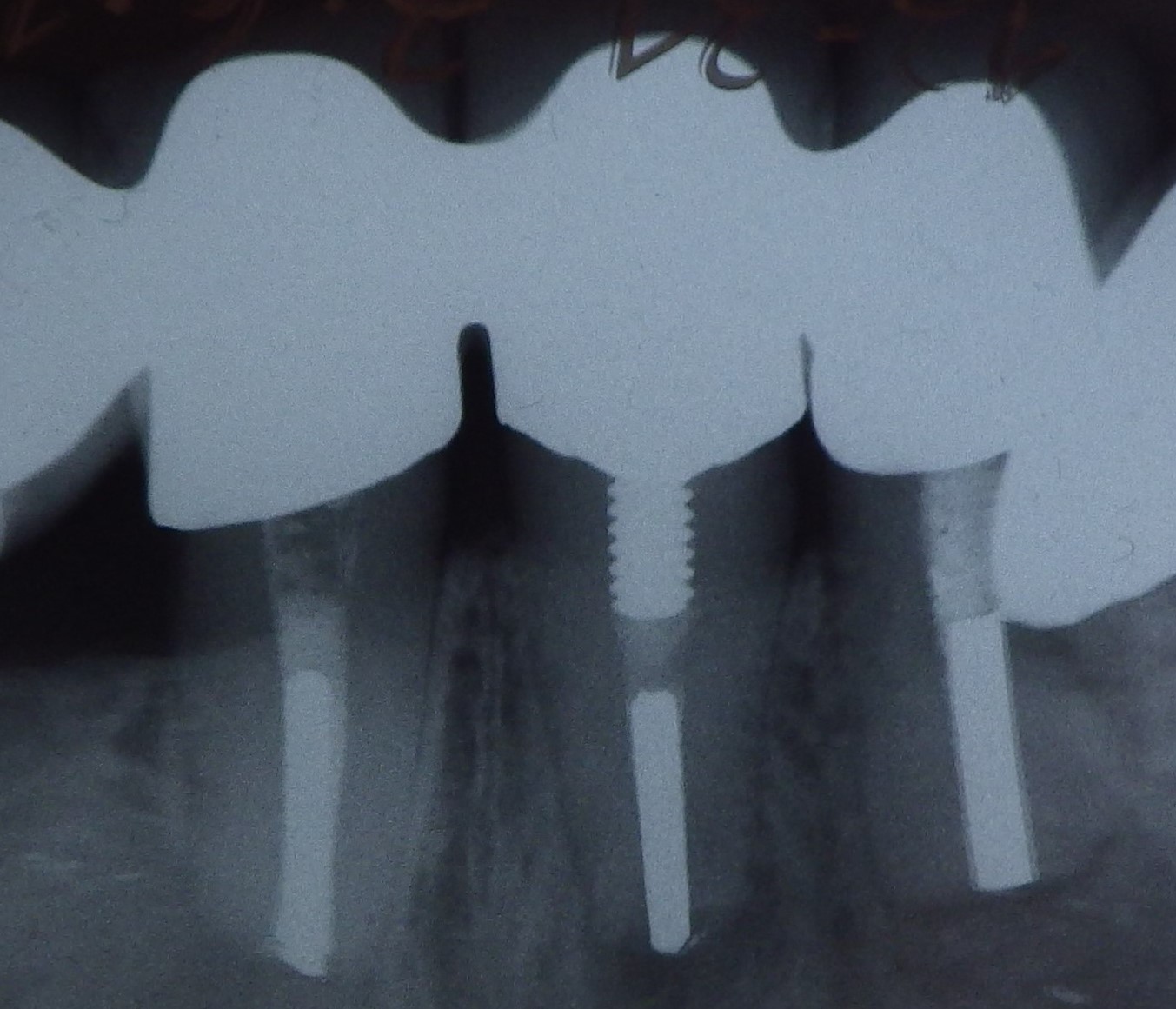 Three different root canal posts
Three different root canal posts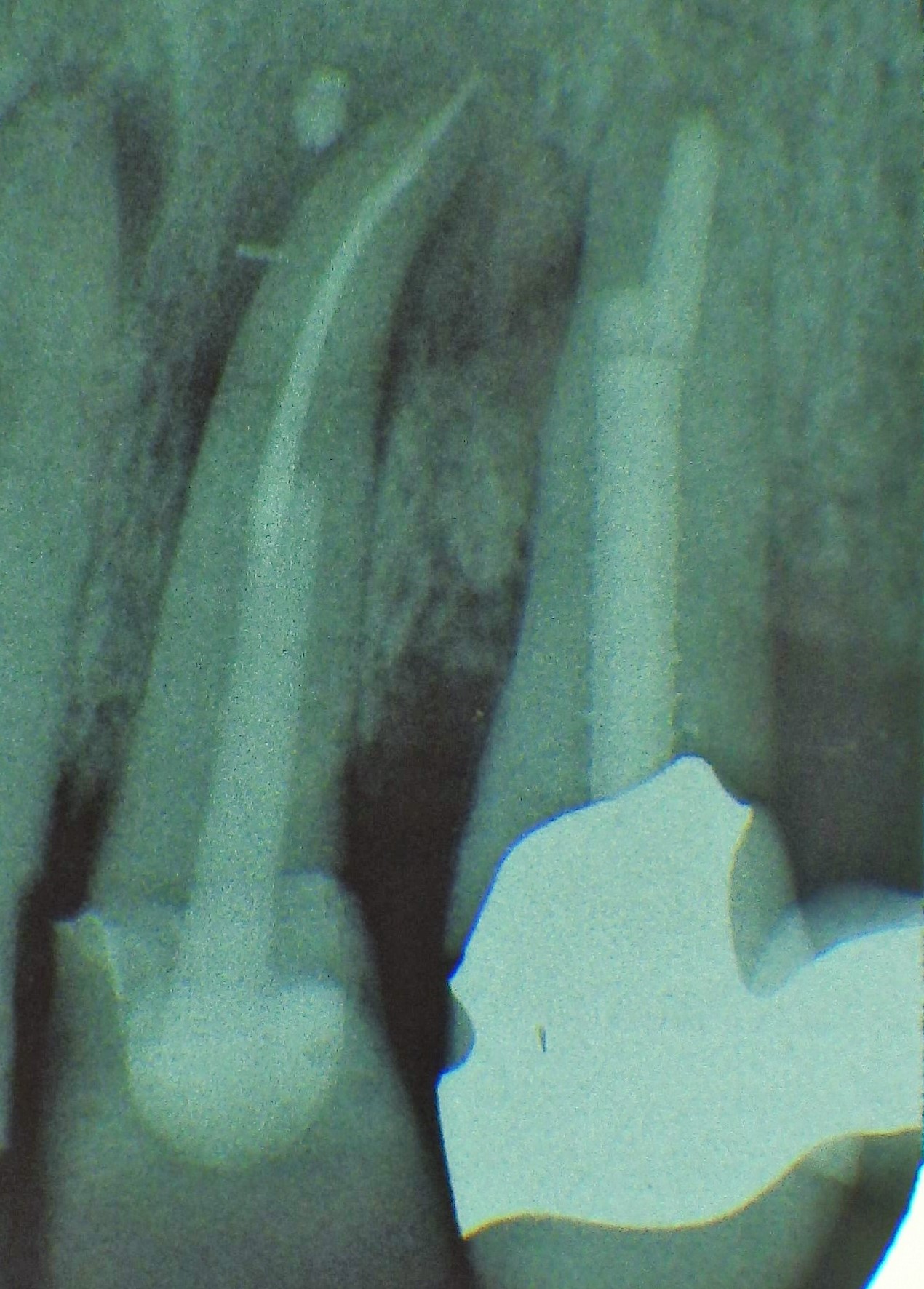 Tooth 14 conical and smooth, tooth 13 cylindrical and threaded
Tooth 14 conical and smooth, tooth 13 cylindrical and threaded